The Thermodynqmics of a Perfect Gas
Examines Boyle's; Charles's; Joule's; Dalton's Laws and details Specific heat of Gasses
Contents
- The Thermodynamics Of A Perfect Gas.
- Boyle's Law.
- Charles's Law.
- The General Gas Equation.
- The Characteristic Gas Equation.
- Pound-mols
- Avrogadro's Hypothesis
- The Universal Gas Equation.
- Joule's Law Of Internal Energy
- The Specific Heats Of A Perfect Gas.
- The Molecular Specific Heat:-
- The Enthalpy Of A Perfect Gas.
- The Ratio Of Specific Heats:-
- Summary
- Page Comments
The Thermodynamics Of A Perfect Gas.
A Perfect Gas is a Working Substance which obeys the Following:- Boyle's Law
- Charles's Law
- Joule's Law of internal Energy
- Dalton's Law of Partial Pressures
- Has a Specific heat which is Constant
- Not be able to change it's state even at Absolute Zero
- The molecules would need to be so far apart that there are no inter molecular forces and no collisions
Boyle's Law.
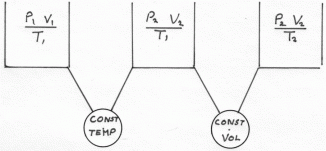
If the Temperature remains constant,. The Pressure of a fixed mass of gas is inversely proportional to it'sVolume.Charles's Law.
If the Volume of a fixed mass of gas is kept Constant, then it's pressure is directly proportional to it's Absolute temperaturePound-mols
The Lb-Mol is a measure of weight and equals the molecular weight of a gas expressed in pounds. One Lb-Mol of Oxygen weighs 32 Lbs. One Lb-Mol of Nitrogen weighs 28 Lbs. One Lb-Mol ofAvrogadro's Hypothesis
This states that equal volumes of gas at the same temperature and Pressure contain the same number of Molecules. This means that one Lb-Mol of ANY gas will occupy the same Volume at the same temperature and Pressure. One lb-Mol at 14.7 psi and atThe Universal Gas Equation.
The universal gas equation ties together pressure, volume and temperature: where R is the universal gas constant. Multiply both sides by the Molecular weight M Note. Mv is a constant for ANY gas at a given temperature and Pressure. Therefore MR is also a Constant for any gas at a given temperature and Pressure. MR is called the universal gas constant and is given the symbolJoule's Law Of Internal Energy
Joule's Law states that for a PERFECT gas the Internal Energy is independent of the Pressure and Volume and depends only on the Temperature of the Gas.The Specific Heats Of A Perfect Gas.
The Specific heat is the quantity of heat required to raise one pound of a substance through a temperature rise of one degree. In the case of gases the Specific Heat depends upon the way in which the gas is heated. i.e. If it is allowed to do work the Specific Heat must be greater. There are therefore an infinite number of Specific Heats but only two will be considered here.- Specific heat at Constant Volume. (C_V).
- Specific heat at Constant Pressure. (C_p)
 Login
Login