Air Compressors
Examines the application of the gas laws to Air Compressors and Motors.
Contents
Introduction
Single Stage Reciprocating without Clearance:MISSING IMAGE!
23287/Air-Compressors-0020.png cannot be found in /users/23287/Air-Compressors-0020.png. Please contact the submission author.
- Suction Stroke: The inlet valve opens and the cylinder fills with air at Ambient Pressure.
- Compression Stroke: Both Valves are shut. The Pressure is raised from
to
.
- Delivery Stage: The Exhaust Valve opens. Air at
is delivered to the receiver at Constant Pressure.
A Comparison Of The Work Done With Different Indices Of Compression
MISSING IMAGE!
23287/Air-Compressors-0022.png cannot be found in /users/23287/Air-Compressors-0022.png. Please contact the submission author.
- 2 Isothermal. n = 1
- 2' Compression when
- 2''
- Adiabatic Reversible.
- 2'''
For Reciprocating Compressors:
- The efficiency referred to is the Isothermal case since fairly successful cooling can be achieved.
For Rotary Compressors:
- The Cooling is very difficult and Indices of less than
are never achieved. It is therefore normal to compare the Performance with the Adiabatic reversible case.
The Cooling Of Compressors.
It is usually considered that the heat is given up during the Compression: For a Polytropic Compression. For an Isothermal Compression.Example:
[imperial]
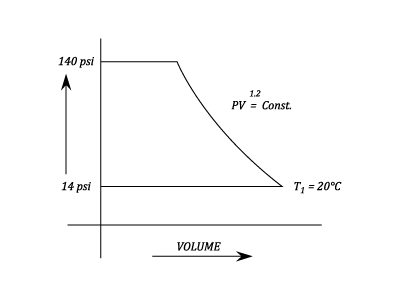
Example - Application of the gas laws to Air Compressors and Motors
Problem
An Air Compressor takes in Air at 14 psi and at 20 degrees C. It is compressed in accord to the law  and delivers it to receiver at 140psi.
Find the Temperature at the end of the Compression and Calculate per pound of Air, the Compressor Work input and the heat rejected during Compression.
and delivers it to receiver at 140psi.
Find the Temperature at the end of the Compression and Calculate per pound of Air, the Compressor Work input and the heat rejected during Compression.
Workings
Solution
Temperature at the end of the Compression  Compressor Work input
Compressor Work input  Heat Rejected during Compression
Heat Rejected during Compression 
 Login
Login